Dmg chemical4/2/2023  
Nickel ion reacts with dimethylglyoxime and forms an insoluble red precipitate of nickel dimethylglyoxime.Its other name is 2,3-Butanedione dioxide. 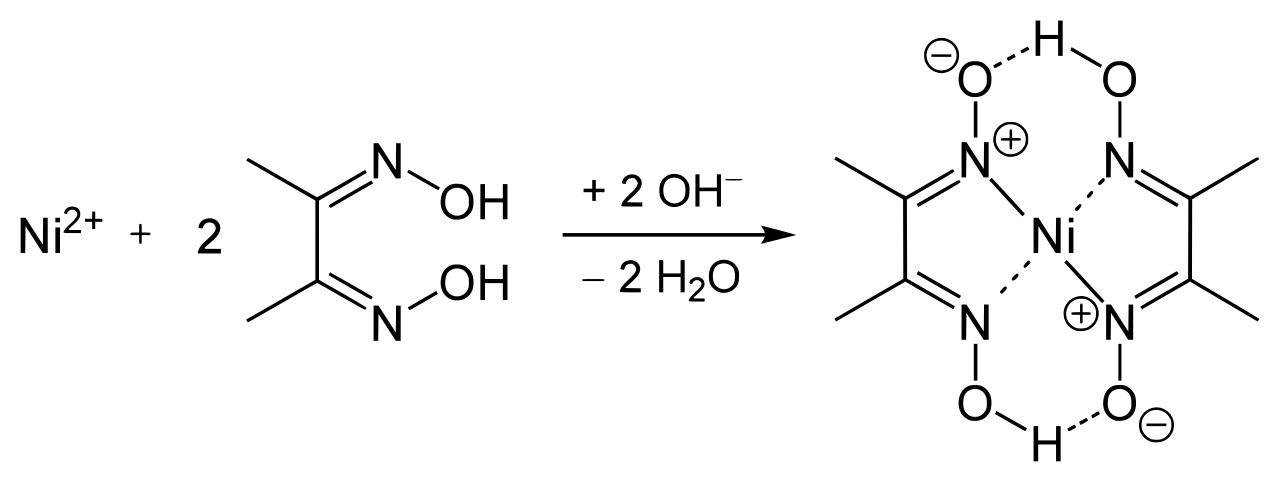
We can prepare many related ligands from other diketones, such as benzil. Its coordination complexes are of theoretical interest as models for enzymes and catalysts both. DmgH2 is useful in the analysis of palladium or nickel.  
This colorless solid is the dioxide derivative of the diketone butane-2,3-dione which is diacetyl. It works as a chelating agent which forms complexes with metals such as copper, nickel, and palladium. Although commonly it is useful as a reagent specific to nickel, Dimethylglyoxime actually allows the detection of other metal ions too. Many derivatives of Dimethylglyoxime are synthesized and proposed. Also, it is very much sensitive and specific reagent for the nickel element. It is one of the first selective organic reagents applied in analytical chemistry. It is soluble in methanol or in sodium hydroxide solution but very less in water. 2 Solved Examples Dimethylglyoxime Formula What is Dimethylglyoxime?ĭimethylglyoxime is a white chemical powder. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
